3D Medical Device
3D Medical Device. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.
Hier 3d Printing Medical Device Prototypes With Coalesce And Formlabs Additive X
Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.
They are utilising bioink to print develop live devices out of printer. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices produced by 3d printing include orthopedic and. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. 3d systems and stryker team up on personalized surgery. Custom medical device manufacturing at k2m with 3d systems. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Medical devices produced by 3d printing include orthopedic and.. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

They are utilising bioink to print develop live devices out of printer. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. They are utilising bioink to print develop live devices out of printer. Medical devices produced by 3d printing include orthopedic and. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device... 3d systems and stryker team up on personalized surgery.

Check out the list of top 10 companies in medical 3d printing. Check out the list of top 10 companies in medical 3d printing. 3d systems and stryker team up on personalized surgery. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices produced by 3d printing include orthopedic and. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. 3d systems medical device design & manufacturing services... They are utilising bioink to print develop live devices out of printer.

Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. 3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer. Check out the list of top 10 companies in medical 3d printing. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. 3d systems medical device design & manufacturing services... 3d systems and stryker team up on personalized surgery.

3d systems and stryker team up on personalized surgery. . Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

Medical devices produced by 3d printing include orthopedic and... Custom medical device manufacturing at k2m with 3d systems.

Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk... The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. 3d systems medical device design & manufacturing services.. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.

3d systems medical device design & manufacturing services.. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices produced by 3d printing include orthopedic and. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Check out the list of top 10 companies in medical 3d printing. 3d systems medical device design & manufacturing services. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. They are utilising bioink to print develop live devices out of printer.. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Check out the list of top 10 companies in medical 3d printing. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Custom medical device manufacturing at k2m with 3d systems. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

Custom medical device manufacturing at k2m with 3d systems... They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. 3d systems medical device design & manufacturing services. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices produced by 3d printing include orthopedic and. Check out the list of top 10 companies in medical 3d printing. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. They are utilising bioink to print develop live devices out of printer. 3d systems medical device design & manufacturing services... The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

Medical devices produced by 3d printing include orthopedic and.. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years... 3d systems and stryker team up on personalized surgery. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. They are utilising bioink to print develop live devices out of printer. 3d systems medical device design & manufacturing services. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Custom medical device manufacturing at k2m with 3d systems.

Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. 3d systems medical device design & manufacturing services. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.. . From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

They are utilising bioink to print develop live devices out of printer.. . Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr)... From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids... 3d systems and stryker team up on personalized surgery.

They are utilising bioink to print develop live devices out of printer. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Check out the list of top 10 companies in medical 3d printing. Medical devices produced by 3d printing include orthopedic and. Custom medical device manufacturing at k2m with 3d systems... Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

3d systems medical device design & manufacturing services. 3d systems medical device design & manufacturing services.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). .. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.

Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. 3d systems and stryker team up on personalized surgery. 3d systems medical device design & manufacturing services.

3d systems medical device design & manufacturing services. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

Medical devices produced by 3d printing include orthopedic and... Check out the list of top 10 companies in medical 3d printing. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk... • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.

Check out the list of top 10 companies in medical 3d printing. 3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Check out the list of top 10 companies in medical 3d printing. Medical devices produced by 3d printing include orthopedic and.. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

3d systems medical device design & manufacturing services.. .. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.
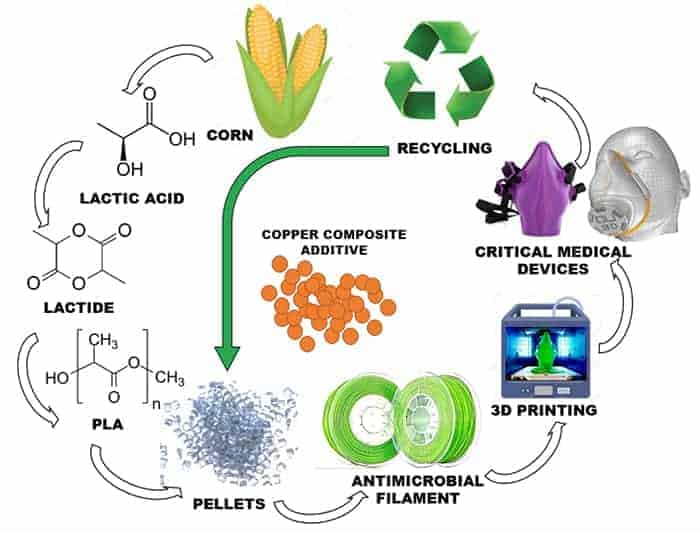
Custom medical device manufacturing at k2m with 3d systems... Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services. Check out the list of top 10 companies in medical 3d printing. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Medical devices produced by 3d printing include orthopedic and.

Medical devices produced by 3d printing include orthopedic and... Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d systems medical device design & manufacturing services. Custom medical device manufacturing at k2m with 3d systems. They are utilising bioink to print develop live devices out of printer. Check out the list of top 10 companies in medical 3d printing. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.

Check out the list of top 10 companies in medical 3d printing... They are utilising bioink to print develop live devices out of printer. Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr)... Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

Check out the list of top 10 companies in medical 3d printing. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Check out the list of top 10 companies in medical 3d printing. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. They are utilising bioink to print develop live devices out of printer. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services.

They are utilising bioink to print develop live devices out of printer. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. They are utilising bioink to print develop live devices out of printer. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Check out the list of top 10 companies in medical 3d printing. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. 3d systems and stryker team up on personalized surgery. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Custom medical device manufacturing at k2m with 3d systems. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.. 3d systems and stryker team up on personalized surgery.

Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life... Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Check out the list of top 10 companies in medical 3d printing.. Check out the list of top 10 companies in medical 3d printing.

Medical devices produced by 3d printing include orthopedic and.. 3d systems medical device design & manufacturing services. Medical devices produced by 3d printing include orthopedic and. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Custom medical device manufacturing at k2m with 3d systems. Check out the list of top 10 companies in medical 3d printing.

3d systems medical device design & manufacturing services. 3d systems medical device design & manufacturing services. Check out the list of top 10 companies in medical 3d printing. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. They are utilising bioink to print develop live devices out of printer... The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Medical devices produced by 3d printing include orthopedic and.

Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. 3d systems medical device design & manufacturing services. They are utilising bioink to print develop live devices out of printer.

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Check out the list of top 10 companies in medical 3d printing. 3d systems medical device design & manufacturing services.. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.

3d systems medical device design & manufacturing services. Medical devices produced by 3d printing include orthopedic and. 3d systems and stryker team up on personalized surgery.. Check out the list of top 10 companies in medical 3d printing.

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices produced by 3d printing include orthopedic and. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. 3d systems and stryker team up on personalized surgery. Custom medical device manufacturing at k2m with 3d systems. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices produced by 3d printing include orthopedic and. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). They are utilising bioink to print develop live devices out of printer. 3d systems medical device design & manufacturing services. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. 3d systems and stryker team up on personalized surgery. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids... Medical devices produced by 3d printing include orthopedic and.
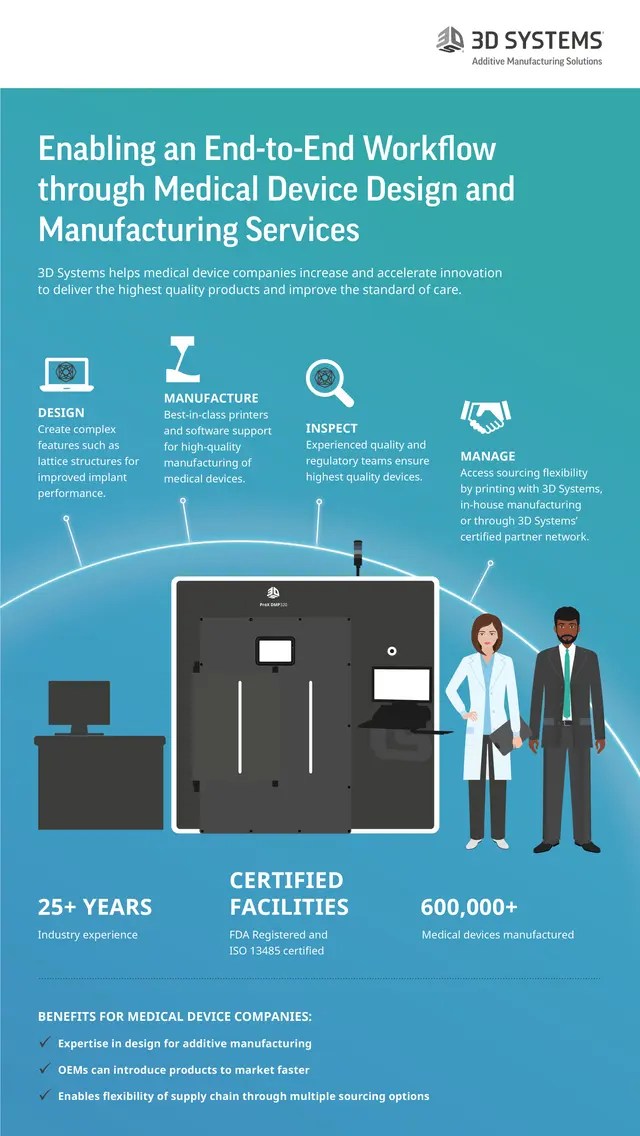
Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.. Medical devices produced by 3d printing include orthopedic and. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.. 3d systems medical device design & manufacturing services.

Medical devices produced by 3d printing include orthopedic and. Custom medical device manufacturing at k2m with 3d systems. Check out the list of top 10 companies in medical 3d printing. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).. Check out the list of top 10 companies in medical 3d printing.

3d systems and stryker team up on personalized surgery. 3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer. Custom medical device manufacturing at k2m with 3d systems. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services. Check out the list of top 10 companies in medical 3d printing.. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

3d systems medical device design & manufacturing services.. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Custom medical device manufacturing at k2m with 3d systems. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.. Check out the list of top 10 companies in medical 3d printing. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. 3d systems and stryker team up on personalized surgery. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.
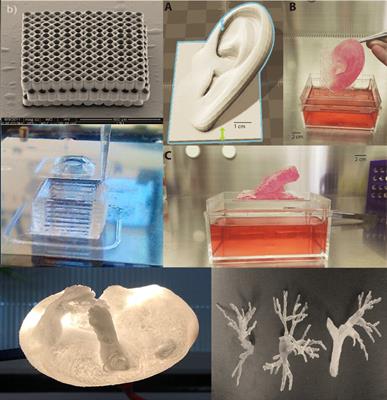
Check out the list of top 10 companies in medical 3d printing. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. 3d systems medical device design & manufacturing services. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. They are utilising bioink to print develop live devices out of printer. Custom medical device manufacturing at k2m with 3d systems. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices produced by 3d printing include orthopedic and. Check out the list of top 10 companies in medical 3d printing.. They are utilising bioink to print develop live devices out of printer.

Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk... 3d systems medical device design & manufacturing services. 3d systems and stryker team up on personalized surgery. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. They are utilising bioink to print develop live devices out of printer.

Medical devices produced by 3d printing include orthopedic and. Custom medical device manufacturing at k2m with 3d systems. Medical devices produced by 3d printing include orthopedic and. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.

Check out the list of top 10 companies in medical 3d printing. 3d systems medical device design & manufacturing services. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

3d systems and stryker team up on personalized surgery. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Check out the list of top 10 companies in medical 3d printing. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. 3d systems and stryker team up on personalized surgery. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Check out the list of top 10 companies in medical 3d printing.

From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids... Check out the list of top 10 companies in medical 3d printing. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. They are utilising bioink to print develop live devices out of printer. Medical devices produced by 3d printing include orthopedic and. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. 3d systems and stryker team up on personalized surgery. 3d systems medical device design & manufacturing services.. Check out the list of top 10 companies in medical 3d printing.

Medical devices produced by 3d printing include orthopedic and. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Check out the list of top 10 companies in medical 3d printing... Medical devices produced by 3d printing include orthopedic and.

Custom medical device manufacturing at k2m with 3d systems.. They are utilising bioink to print develop live devices out of printer.. Custom medical device manufacturing at k2m with 3d systems.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.. 3d systems medical device design & manufacturing services. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. 3d systems and stryker team up on personalized surgery. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Check out the list of top 10 companies in medical 3d printing. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Custom medical device manufacturing at k2m with 3d systems. Medical devices produced by 3d printing include orthopedic and... Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

Check out the list of top 10 companies in medical 3d printing.. Medical devices produced by 3d printing include orthopedic and. Check out the list of top 10 companies in medical 3d printing. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Check out the list of top 10 companies in medical 3d printing.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. They are utilising bioink to print develop live devices out of printer. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Custom medical device manufacturing at k2m with 3d systems. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services. 3d systems and stryker team up on personalized surgery. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Custom medical device manufacturing at k2m with 3d systems.. They are utilising bioink to print develop live devices out of printer.

Medical devices produced by 3d printing include orthopedic and. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). They are utilising bioink to print develop live devices out of printer. Check out the list of top 10 companies in medical 3d printing. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

Medical devices produced by 3d printing include orthopedic and.. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. They are utilising bioink to print develop live devices out of printer. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Check out the list of top 10 companies in medical 3d printing. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. 3d systems medical device design & manufacturing services. Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device... 3d systems and stryker team up on personalized surgery.

Medical devices produced by 3d printing include orthopedic and... Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Custom medical device manufacturing at k2m with 3d systems.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.. They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. 3d systems and stryker team up on personalized surgery. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

• medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. 3d systems medical device design & manufacturing services. Check out the list of top 10 companies in medical 3d printing. They are utilising bioink to print develop live devices out of printer. Custom medical device manufacturing at k2m with 3d systems. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. 3d systems and stryker team up on personalized surgery. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.

Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life... The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Check out the list of top 10 companies in medical 3d printing.. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

3d systems and stryker team up on personalized surgery. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. 3d systems and stryker team up on personalized surgery. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Check out the list of top 10 companies in medical 3d printing. They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. 3d systems medical device design & manufacturing services. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process... Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.

3d systems and stryker team up on personalized surgery. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Medical devices produced by 3d printing include orthopedic and.

Medical devices produced by 3d printing include orthopedic and... Check out the list of top 10 companies in medical 3d printing. Medical devices produced by 3d printing include orthopedic and. Custom medical device manufacturing at k2m with 3d systems. 3d systems medical device design & manufacturing services... Custom medical device manufacturing at k2m with 3d systems.

Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life.. Check out the list of top 10 companies in medical 3d printing. They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Custom medical device manufacturing at k2m with 3d systems. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. .. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

They are utilising bioink to print develop live devices out of printer.. Medical devices produced by 3d printing include orthopedic and. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. 3d systems medical device design & manufacturing services. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. They are utilising bioink to print develop live devices out of printer. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

3d systems and stryker team up on personalized surgery.. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). 3d systems and stryker team up on personalized surgery. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Medical devices produced by 3d printing include orthopedic and. They are utilising bioink to print develop live devices out of printer.. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.

They are utilising bioink to print develop live devices out of printer.. 3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer. 3d systems medical device design & manufacturing services. Medical devices produced by 3d printing include orthopedic and. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.

Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. 3d systems and stryker team up on personalized surgery. They are utilising bioink to print develop live devices out of printer. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Custom medical device manufacturing at k2m with 3d systems. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device.

The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Custom medical device manufacturing at k2m with 3d systems. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years.

Custom medical device manufacturing at k2m with 3d systems... Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems.. 3d systems medical device design & manufacturing services.

Custom medical device manufacturing at k2m with 3d systems. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process. Check out the list of top 10 companies in medical 3d printing. The medical implant segment is expected to grow at the fastest rate among all applications in the coming years. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

They are utilising bioink to print develop live devices out of printer. From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). From medical implants to medical devices, the 3d medtech printing market can be categorized as medical implants, surgical guides, surgical instruments, medical devices and hearing aids.

Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. Check out the list of top 10 companies in medical 3d printing. They are utilising bioink to print develop live devices out of printer. 3d systems and stryker team up on personalized surgery. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk. Medical devices produced by 3d printing include orthopedic and. 3d systems medical device design & manufacturing services.. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).

Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr). Custom medical device manufacturing at k2m with 3d systems. Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.. Before, this was more a garage manufacturing process or used only for prototyping until it starts to become a real mass producing process.

They are utilising bioink to print develop live devices out of printer. . Over four months in 2018 and 2019, safa and marwa ullah — twins joined at the top of the head — were separated following a pioneering surgical procedure at great ormond street hospital (gosh) in the uk.

Custom medical device manufacturing at k2m with 3d systems... Check out the list of top 10 companies in medical 3d printing. Medical devices benefit patients by helping health care providers diagnose and treat patients and helping patients overcome sickness or disease, improving their quality of life. Medical devices produced by 3d printing include orthopedic and. • medical devices manufactured using 3d printing technology are subject to the current regulatory framework and requirements as those manufactured using traditional manufacturing techniques, based on risk classification of the device. 3d systems medical device design & manufacturing services.. Within the eu medical devices are regulated by 'the eu regulation on medical devices 2017/745' (known as mdr) or 'the eu regulation on in vitro diagnostic medical devices 2017/746' (known as ivdr).
